
PESG Research Report: Revolutionary Surgical Infection Prevention: PolyPid’s D-PLEX100 Approaches Critical Phase 3 Data Readout
New report from PESG Research brand covers PolyPid*, which is tackleing the $10 billion dollar a year surgeical site infections cost with its innovative PLEX technology
/EIN News/ -- New York, May 30, 2025 (GLOBE NEWSWIRE) -- PESG Research is releasing a report today examining PolyPid Ltd.*, an innovative late-stage biopharmaceutical company developing revolutionary localized drug delivery technologies for surgical infection prevention. The below report explores the company's breakthrough PLEX platform technology, its upcoming topline phase 3 data, and potential implications for transforming surgical care practices in the multi-billion dollar surgical site infection prevention market.
This report contains sponsored content, please see refer to the disclaimers and disclosures included at the end of this report.
Executive Summary
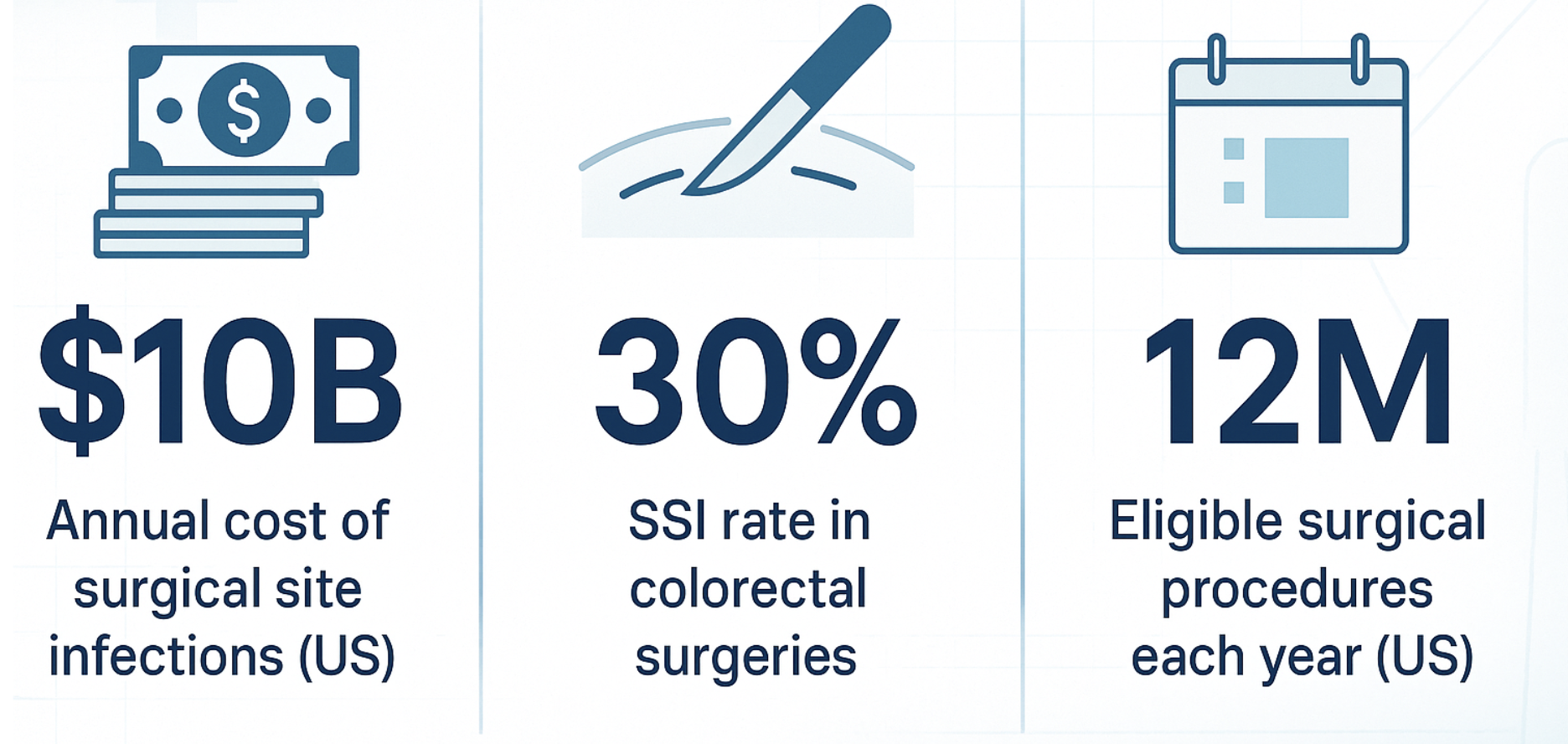
The surgical site infection (SSI) prevention landscape stands at a potential inflection point as PolyPid Ltd. prepares to announce topline results from its pivotal SHIELD II Phase 3 trial by the end of Q2 2025. The company's D-PLEX100 represents a paradigm shift in infection prevention, utilizing novel polymer-lipid encapsulation matrix (PLEX) technology to deliver sustained antibiotic release directly at surgical sites for 30 days. With SSIs affecting up to 30% of colorectal surgeries and imposing substantial healthcare costs estimated at $10 billion annually in the US and EU, successful development of D-PLEX100 could address a critical unmet medical need. The FDA's assignment of Breakthrough Therapy, Fast Track, and Qualified Infectious Disease Product (QIDP) designations underscores the regulatory recognition of this approach's potential significance.
The Critical Challenge of Surgical Site Infections
Surgical site infections remain one of healthcare's most persistent challenges, representing 20% of all healthcare-associated infections in US hospitals. Despite advances in surgical techniques and prophylactic protocols, SSI rates in high-risk procedures like colorectal surgery can reach 30%, leading to extended hospital stays of 7-11 additional days and mortality risk increases of 2-11 fold. The economic burden is staggering, with direct costs averaging $11,000-26,000 per infection and total annual costs reaching approximately $10 billion in the US alone.
Current prevention strategies rely primarily on systemic antibiotic prophylaxis administered intravenously 30-60 minutes before surgery. However, this approach faces fundamental limitations. Surgical incisions disrupt local blood flow, significantly limiting antibiotic penetration to the precise site where infections occur. Moreover, systemic administration exposes patients to higher drug concentrations throughout the body, potentially increasing toxicity risks and contributing to antimicrobial resistance development.
PLEX Technology: A Novel Approach to Localized Drug Delivery
PolyPid's proprietary PLEX technology represents a sophisticated advancement in controlled drug delivery systems. The platform creates thousands of alternating layers of biocompatible polymers and lipids that physically embed active pharmaceutical ingredients. As these outer layers gradually disintegrate upon exposure to body fluids, they enable precise, predetermined drug release rates spanning several days to months.
For D-PLEX100, this technology pairs with doxycycline, a broad-spectrum antibiotic effective against both gram-positive and gram-negative bacteria, including methicillin-resistant Staphylococcus aureus (MRSA) and other antibiotic-resistant strains. The formulation achieves local antibiotic concentrations 10-115 times higher than systemic administration while using only a fraction of the total drug amount, potentially minimizing systemic exposure and associated side effects.
Clinical Development Progress and Regulatory Recognition
The clinical development program for D-PLEX100 has demonstrated encouraging signals across multiple surgical contexts. In Phase 2 abdominal surgery trials, D-PLEX100 plus standard of care achieved a statistically significant 59% reduction in the primary endpoint compared to standard of care alone (p=0.0086). Perhaps more notably, the treatment group experienced zero deaths compared to five in the control arm within 60 days post-surgery.
The pivotal SHIELD I Phase 3 trial, while not meeting its primary endpoint in the full intent-to-treat population, revealed compelling efficacy in a pre-specified subgroup analysis. Among patients with large surgical incisions (>20 cm), D-PLEX100 demonstrated a statistically significant 54% reduction in the composite primary endpoint of SSIs, reinterventions, and mortality (p=0.0032). This finding directly informed the design of the ongoing SHIELD II trial, which focuses specifically on this higher-risk patient population.
The FDA's regulatory designations reflect recognition of D-PLEX100's potential significance. Breakthrough Therapy designation is reserved for drugs that demonstrate substantial improvement over existing treatments for serious conditions. Fast Track designation facilitates more frequent FDA communications and potentially accelerated review timelines. The QIDP designation provides additional market exclusivity incentives for addressing antimicrobial resistance challenges.
SHIELD II: A Potentially Definitive Study
The ongoing SHIELD II Phase 3 trial represents a carefully designed study incorporating lessons learned from SHIELD I. Following an independent Data Safety Monitoring Board's review of unblinded efficacy data from the first 430 enrolled patients, the board recommended concluding the study at 800 patients—the lowest sample size reassessment option available. This recommendation, while requiring additional enrollment beyond the initially planned 624 patients, may suggest positive efficacy trends in the analyzed interim data.
The study's multinational design spans approximately 60 centers across the United States, Europe, and Israel, enhancing the generalizability of results across different healthcare settings and patient populations. The primary endpoint focuses on a composite of SSI events, reinterventions, and mortality within 30 days post-surgery, as adjudicated by an independent committee.
Broader Industry Implications
Success of D-PLEX100 could catalyze broader adoption of localized drug delivery approaches in surgical settings. The technology's platform nature suggests potential applications beyond infection prevention, including localized chemotherapy delivery through the company's OncoPLEX program, currently in preclinical development for solid tumor treatment.
The substantial market opportunity—with approximately 12 million eligible procedures annually in the US and 8 million in Europe—underscores the potential healthcare impact. PolyPid has already secured European commercialization rights through a partnership with Advanz Pharma, valued at up to $115 million plus royalties, while advancing discussions for US market partnerships.
Conclusion
As the pharmaceutical industry grapples with antimicrobial resistance and healthcare systems seek cost-effective solutions to persistent clinical challenges, D-PLEX100's approaching data readout represents a potentially significant milestone. The convergence of compelling preclinical and early clinical data, regulatory recognition, and substantial unmet medical need positions this development as one to monitor closely in the evolving landscape of surgical infection prevention.
Recent News Highlight from PolyPid: PolyPid Announces Successful Completion of Enrollment in Phase 3 SHIELD II Trial of D-PLEX₁₀₀ for the Prevention of Abdominal Colorectal Surgical Site Infections
>> Click here to Subscribe for more updates like this or go to https://www.pesgresearch.com/subscribe
* Legal Disclaimer & Disclosure:
Nothing in this report constitutes medical, financial or any form professional or licensedadvice. This report is published by ‘PESG Research’, a digital promotional content brand who’s operators are compensated to provide digestable and favorabel coverage of companies. This report contains and is a form of paid promotional content or advertising for PolyPid Ltd and was produced as part of the fee's they pay PESG's operators, Arx Advisory Ltd. This report has not been reviewed or approved by PolyPId prior to publication and it does not represent an official communication from PolyPid. The operators of PESG Research received or are expected to receive a monthly recurring fee of five thousand united states dollars via wire transfer from PolyPid as part of an ongoing agreement starting May 1, 2025 in return for distribution and promotional coverage services, and receive additional monthly compensation for non promotional unrelated data and advisory services on top of that. Please review the full disclaimers and compensation disclosures here for further details: redditwire.com/terms. Readers are advised to refer to the official materials of the company aforementioned. The report should not be treated as objective.

Distribution channels: Media, Advertising & PR ...
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
Submit your press release

